Manténgase sano!
Mantenerse informado(a) promueve la buena salud. Manténgase al día con las últimas noticias médicas encontradas aquí.
09 Jun
New Study Suggests No Major Adverse Outcomes With Early GLP-1 Exposure During Pregnancy
Researchers found no substantial increase in the risk of pregnancy loss, abnormal fetal growth, or major congenital malformations among pregnancies with first-trimester GLP-1 exposure. But they say more studies are needed.
08 Jun
Study Finds Just 5 Minutes of Prayer Helps Reduce Pain and Anxiety
Five-minute in-person intercessory prayer sessions can significantly reduce pain and anxiety in patients, according to new research.
05 Jun
How Brain Surgery Helped an Illinois Mom Grow Her Family
When medication stopped working and a brain tumor stood in the way of another pregnancy, one Illinois mom faced a life-changing decision.
Teen Recovering From Concussion? A 'Sweet Spot' For Screen Time Could Speed Up Their Recovery
Moderating a teen’s screen time following a concussion might help speed their recovery, a new study says.
Teenagers who kept their screen time to around 141 minutes a day within the first three days of their concussion sped up their recovery by about 35%, researchers reported June 9 in the British Journal of Sports Medicine....
- Dennis Thompson HealthDay Reporter
- |
- June 10, 2026
- |
- Página completa
Retro Video Game Aids Stroke Recovery, Improves Arm Function
A customized throwback video game might help stroke survivors regain arm function, a new study says.
The '90s-style video game requires players to use their arm muscles to complete tasks like flying a helicopter around the screen to hit a moving target, researchers reported June 8 in the journal Neurorehabilitation and Neural Repair
- Dennis Thompson HealthDay Reporter
- |
- June 10, 2026
- |
- Página completa
Women Hit Harder By Sleep Apnea Than Men, Study Finds
Women with sleep apnea tend to suffer from it more than men, even though they wake in the night about as often, a new study says.
Women reported much higher levels of headache, nightmares and needing to go to the bathroom at night due to their sleep apnea, researchers will report at the upcoming annual meeting of the American Academy of Sl...
- Dennis Thompson HealthDay Reporter
- |
- June 10, 2026
- |
- Página completa
Once-Daily GLP-1 Pill May Offer New Option for Weight Loss, Diabetes
Another GLP-1 pill could be headed for the market soon, according to early clinical trial results.
The once-daily experimental pill elecoglipron helped people control their blood sugar and lose weight in a pair of phase 2 clinical trials, researchers reported June 8 in The Lancet.
Based on these results, elecoglipron will ad...
- Dennis Thompson HealthDay Reporter
- |
- June 10, 2026
- |
- Página completa
How Much Alcohol Is Actually Safe? A New Study Challenges Old Advice
The debate on "healthy drinking" has shifted again, according to a study released independently today, after the Trump administration decided not to include its findings in new dietary guidelines.
The study — published June 8 in the Journal of Studies on Alcohol and Drugs — concluded that even one drink a day increases...
- Andria Park Huynh HealthDay Reporter
- |
- June 9, 2026
- |
- Página completa
Air Pollution Might Contribute To Clogged Arteries, Heart Disease Risk
Long-term exposure to air pollution might contribute to clogged arteries and heart disease, a new study says.
People with higher levels of exposure to smog had an elevated risk of calcium deposits and plaques in their arteries, researchers reported today in the journal Radiology.
In particular, women had an 81% increased ris...
- Dennis Thompson HealthDay Reporter
- |
- June 9, 2026
- |
- Página completa
New Study Suggests No Major Adverse Outcomes With Early GLP-1 Exposure During Pregnancy
New research is shedding light on first-trimester GLP-1 exposure during pregnancy — and the findings may offer some reassurance.
GLP-1 use among women of reproductive age is increasing. And while current recommendations call for discontinuation before pregnancy, unintentional early exposure can occur.
The medications, which in...
- HealthDay Staff HealthDay Reporter
- |
- June 9, 2026
- |
- Página completa
Feeding Babies Eggs Sooner May Cut Allergy Risk, Study Suggests
Feeding babies egg early in their lives can lead to a decline in egg allergies, particularly among infants with eczema, a new study says.
Cases of egg allergy decreased in Australia within a decade of the nation adopting guidelines that recommend the introduction of egg to infants around 6 months of age, researchers reported June 8 in ...
- Dennis Thompson HealthDay Reporter
- |
- June 9, 2026
- |
- Página completa
Infections A ‘Major Health Hazard’ For People With Diabetes, Large Study Warns
Diabetes wreaks havoc on the body, doing damage to the heart, kidneys, eyes and other major organs.
But one of the most important health risks from diabetes has not gotten the attention it deserves, researchers argue.
Infections should be considered a major health hazard for anyone with diabetes, according to a new study.
Peopl...
- Dennis Thompson HealthDay Reporter
- |
- June 9, 2026
- |
- Página completa
Children's Well-Being Plummets Across 29 States, Report Finds
The kids are not all right, at least in the United States, according to a new report showing a nosedive in children's well-being from 2019 to 2024.
In 29 states, the overall U.S. score fell from 553 to 547 on a 1,000-point scale, a decline that surpasses pre-pandemic numbers, the report found.
This score measures children's well-bein...
- Andria Park Huynh HealthDay Reporter
- |
- June 8, 2026
- |
- Página completa
Just 5 Minutes Of Prayer Helps Reduce Pain and Anxiety, Study Finds
Five minutes of prayer may help ease pain and anxiety, according to a new study from the University of Maryland School of Medicine.
"The prayer intervention was effective regardless of the patient’s faith or no faith," said co-author Joshua Brown, a professor at Indiana University and director and co-founder of the Global Medical Res...
- HealthDay Staff HealthDay Reporter
- |
- June 8, 2026
- |
- Página completa
ADHD ‘Masking’ May Help People Blend In But Harms Mental Health
It can be incredibly difficult for adults with ADHD to fit in socially.
But trying to hide the telltale signs of ADHD could cost their mental health and well-being, a study says.
Adults with ADHD might better fit in if they pretend to pay attention, suppress their urge to fidget, rehearse conversations or over-prepare for meetings, r...
- Dennis Thompson HealthDay Reporter
- |
- June 8, 2026
- |
- Página completa
Why Alcohol Makes You Crave Salty Snacks — And How Protein-Rich Foods Can Help Prevent Weight Gain
There’s a biological reason why booze makes a person crave bar snacks like chips, nuts, fries and pizza, a new study argues.
Alcohol appears to trigger a hormone associated with cravings for savory flavors, researchers reported recently in the journal Obesity Reviews.
This hormone, FGF21, is linked to protein appetite ...
- Dennis Thompson HealthDay Reporter
- |
- June 8, 2026
- |
- Página completa
Getting The RSV Shot While Pregnant Could Protect Your Baby After Birth
There’s now real-world evidence that pregnant women who get the RSV vaccine reduce their newborn’s risk of becoming so sick they require hospitalization, a new study says.
Vaccination against respiratory syncytial virus (RSV) reduces the risk of hospitalization in babies by nearly 70%, researchers reported June 5 in JAMA Ne...
- Dennis Thompson HealthDay Reporter
- |
- June 8, 2026
- |
- Página completa
Irregular Sleep Risks Preschool Kids' Brain Power
Irregular sleep is linked to lower brain power among preschool kids, a new study says.
Children who don’t stick to a regular sleep schedule tend to have problems with vocabulary and memory, according to research to be presented at an upcoming meeting of the American Academy of Sleep Medicine and the Sleep Research Society. The findin...
- Dennis Thompson HealthDay Reporter
- |
- June 8, 2026
- |
- Página completa
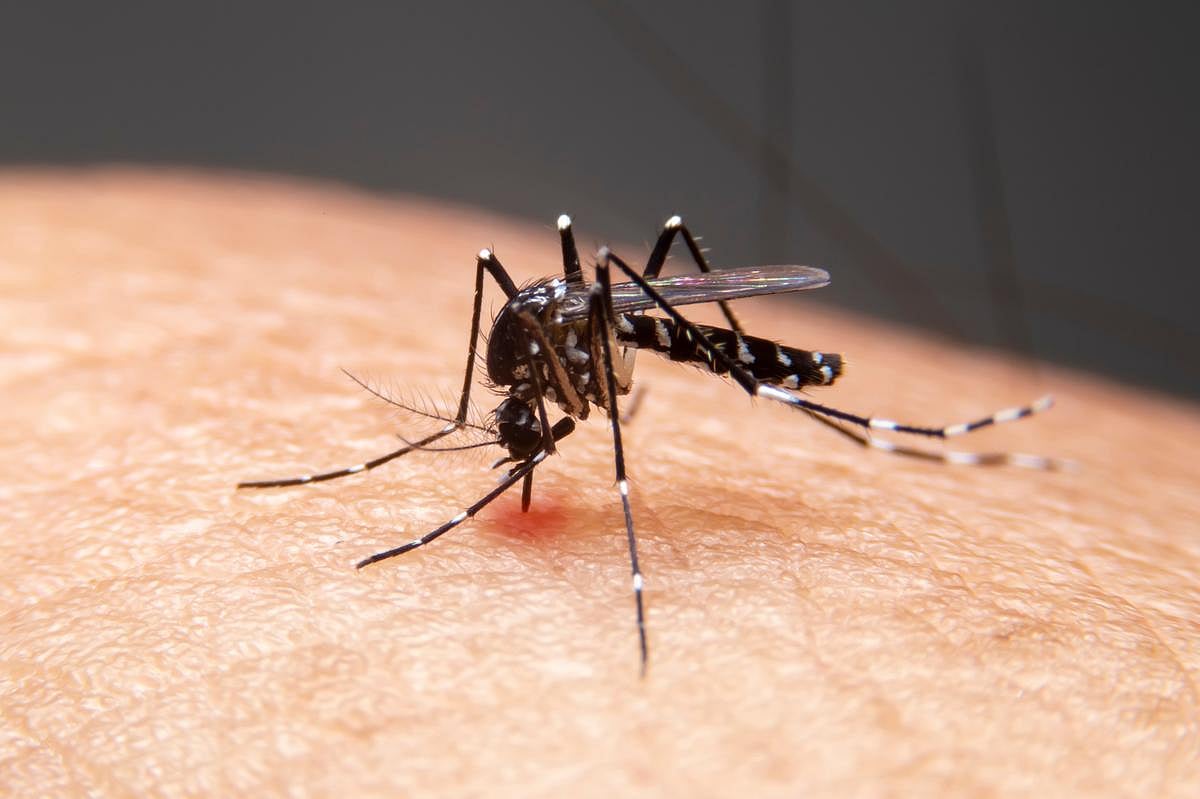
Dengue Is No Longer Just A Travel Risk — What Google’s Mosquito Plan Could Mean For Your Summer
This is not science fiction or some perverse prank.
A Silicon Valley tech giant is seeking federal approval to release up to 64 million sterilized male mosquitoes in California and Florida over the next two years.
The aim?
To drive down the population of Aedes aegypti mosquitoes, which transmit infectious d...
- Andria Park Huynh HealthDay Reporter
- |
- June 5, 2026
- |
- Página completa
Brain Surgery For Pituitary Tumor Helps Illinois Mom Have Second Baby
A suburban Chicago woman who feared she might never have another child is now celebrating a growing family after brain surgery at Northwestern Medicine.
After the birth of her first daughter in 2022, Lisa Fasone began experiencing postpartum hormonal issues.
"My menstrual cycle never came back. After I had my baby and I was done nurs...
- HealthDay Staff HealthDay Reporter
- |
- June 5, 2026
- |
- Página completa
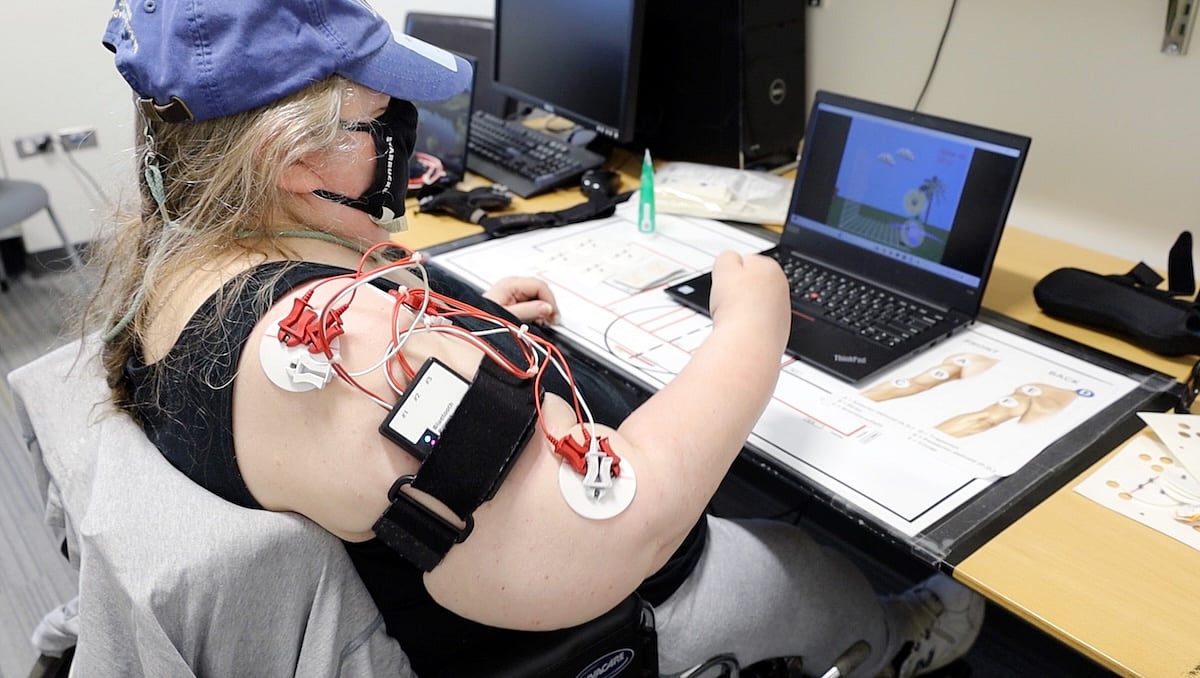
Spinal Cord Stimulation May Restore Arm Strength After Stroke
Electrical stimulation of the spinal cord might improve arm function among people who’ve suffered a stroke, pilot clinical trial results say.
Seven stroke survivors with profound muscle weakness had an average 32% increase in their arm strength after receiving spinal cord stimulation (SCS), researchers reported June 4 in the journal ...
- Dennis Thompson HealthDay Reporter
- |
- June 5, 2026
- |
- Página completa
Popular Blood Pressure Meds Linked To Kidney Damage Risk In Type 2 Diabetes
A common class of blood pressure medications might contribute to kidney damage among people with type 2 diabetes, a new study says.
Dihydropyridine calcium-channel blockers (DCCBs) work by relaxing blood vessels, and are frequently used as second-line therapies in people with diabetic kidney disease (DKD), researchers said.
But patie...
- Dennis Thompson HealthDay Reporter
- |
- June 5, 2026
- |
- Página completa
Too Much Sitting In Pregnancy Doubles Risk Of Complications
In the old days, expecting mothers were encouraged to take it easy as much as possible.
But that advice had it completely backward, a new study says.
There are higher odds of pregnancy complications for women who spend more time sitting, compared to those who include even light activity in their daily routine, researchers reported re...
- Dennis Thompson HealthDay Reporter
- |
- June 5, 2026
- |
- Página completa